The Chemistry of Life
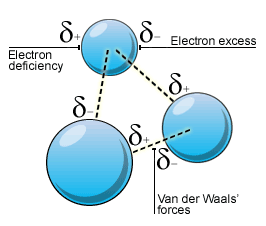
A. Key Forces Acting on Matter
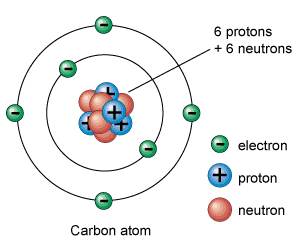
B. Matter1. atoms: a basic unit of matter with a specific number of protons for each electron. Atoms have a neutral charge when isolated. This means that the number of protons will equal the number of electrons in an atom.
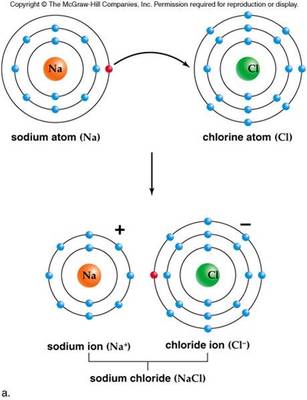
2. elements: consists of all the same type of atoms 3. compounds: 2 or more elements chemically bonded 4. molecules: 2 or more atoms chemically bonded 5. ions: an atom that is charged due to a different number of electrons and protons 6. radioactivity: energy released when atomic nuclei are unstable due to a low number of electrons. 7. isotopes: atoms with the same number of electrons but a different number of neutrons. E. Chemical Bonds |
C. The Atom
The Quantum Mechanical Model of the atom is the current theory of the atom
that states that strong forces bind protons and neutrons together to form the nucleus and the electrons surround the nucleus. D. Sub-Atomic Particles
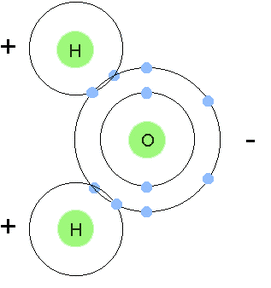
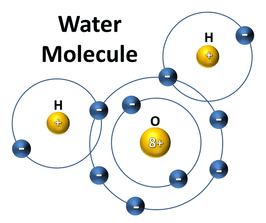
F. Water and Its Importance to Life Water is the most abundant compound in most living things. It contains two hydrogen atoms for every oxygen atom. The positive charges on its 10 protons balance out the negative charge on its 10 electrons.
A polar molecule is a molecule in which the charges are unevenly distributed. The molecule is like a magnet with poles. A water molecule is polar because there is an uneven distribution of electrons between the oxygen and hydrogen atoms. The negative pole is near the oxygen atom and the positive pole is between the hydrogen atoms.
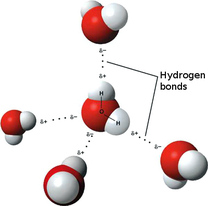
A water molecule can be involved in as many as four hydrogen bonds at a time. Many of water's properties are due to the ability of water to form multiple hydrogen bonds at a time. Cohesion is an attraction between molecules of the same substance. Water is extremely cohesive because of hydrogen bonding. Adhesion occurs when there is an attraction between molecules of different substances. The surface of the water in a graduated cylinder dips slightly in the center because the adhesion between water molecules and glass molecules is stronger than the cohesion between water molecules. All components of a solution are evenly distributed throughout the solution. Salt is the solute in a saltwater solution and water is the solvent-- the substance in which the solute dissolves. Water is the best solvent because its polarity gives it the ability to dissolve both ionic compounds and other polar molecules.
The pH scale measures the concentration of hydrogen ions in a solution. It ranges from 0 to 14. The concentration of H+ ions and OH- ions is equal at a pH of 7. Water has a pH of 7. When solutions are acidic, they have a pH below 7 because they contain more H+ ions than OH- ions. The lower the pH is, the greater the acidity. Solutions with a pH above 7 are called basic because they have more OH- ions than H+ ions. The solution is more basic the higher the pH gets.
Image Sources |