Enzyme-Controlled Reactions
A.- Problem
The purpose of this lab is to observe how enzymes act on substrates to produce products. We will observe how enzyme activity is altered when the enzyme environment's pH is changed.
B.- Hypothesis
E.- Results
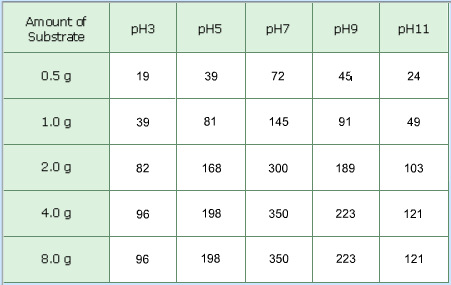
Number of Molecules of Product Formed per Minute (x10^6)
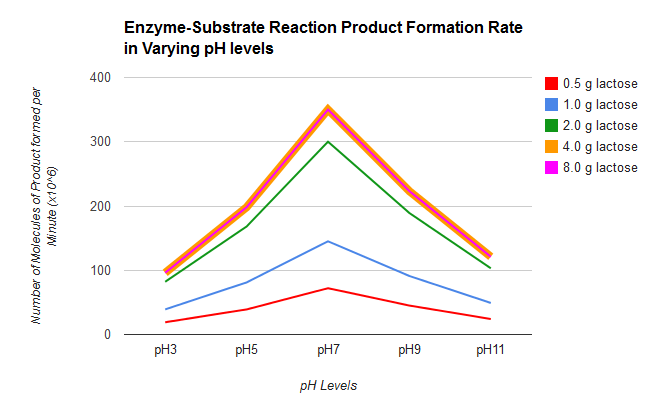
This experiment consisted of adding lactose to five different test tubes which contained a lactase solution in different levels of pH. The tubes had the same levels of pH throughout the experiment: 3, 5, 7, 9, and 11. The variable that changed in the five different trials was the concentration of lactose. 0.5 g, 1.0 g, 2.0 g, 4.0 g, and 8,0 g of substrate were added to the tubes in five different trials The number of molecules formed per minute was always higher with a neutral pH.
F.- Conclusion
Paragraph Conclusion
I had predicted that if the pH of the lactase reaction was more acidic, the products formed would be a lot more than what were normally produced. My hypothesis was disproved by this experiment because the enzymes reacted more effectively in a neutral pH. Enzymes like an optimal pH because pH can alter enzyme shape (denature) and the active site may be destroyed not allowing it to bond with the substrate to form products. My results showed that more product is formed when adding more substrate. In trial four and five, the same results were obtained because there were no more enzymes for the substrate to bond with in the fifth trial. Lactase (enzyme) likes a pH of 7 as evidenced by data showing the highest rate of product formation always being greater at pH 7 for every trial using different concentrations of lactase (substrate). I do not think that this experiment needs any improvements. I did not find any errors in this experiment.
|
C.- Materials
D.- Procedure
.
|